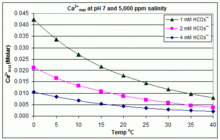
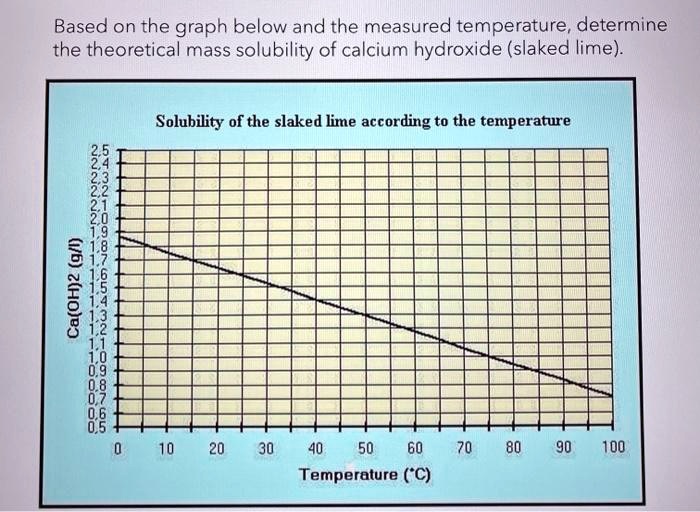
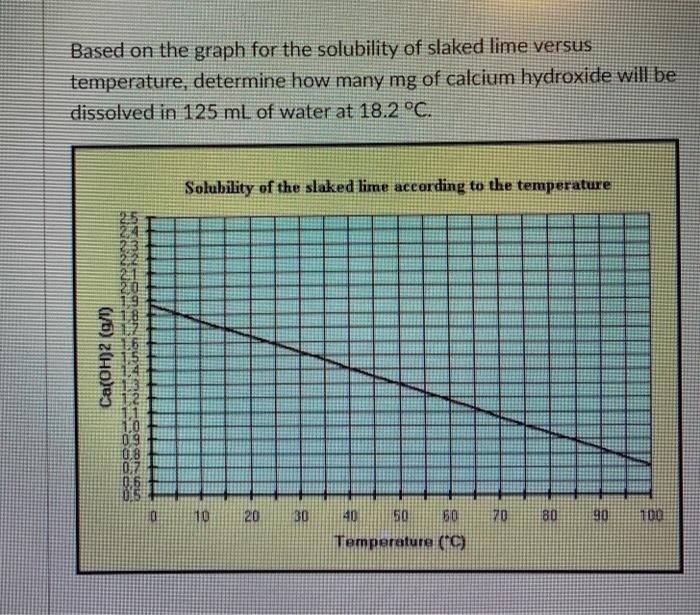
SOLVED:The solubility of slaked lime, Ca(OH)2, in water at 20^∘ C is 0.185 g / 100.0 mL . What volume of 0.00100 M HCl is needed to neutralize 10.0 mL of a saturated Ca(OH)2 solution?

Processes | Free Full-Text | Solubility Data of Potential Salts in the MgO-CaO-SO2-H2O-O2 System for Process Modeling

Solubility of calcium carbonate (lime scale) in water as a function of pH. | Download Scientific Diagram

CLXXII.—The solubility of lime in water - Journal of the Chemical Society, Transactions (RSC Publishing)





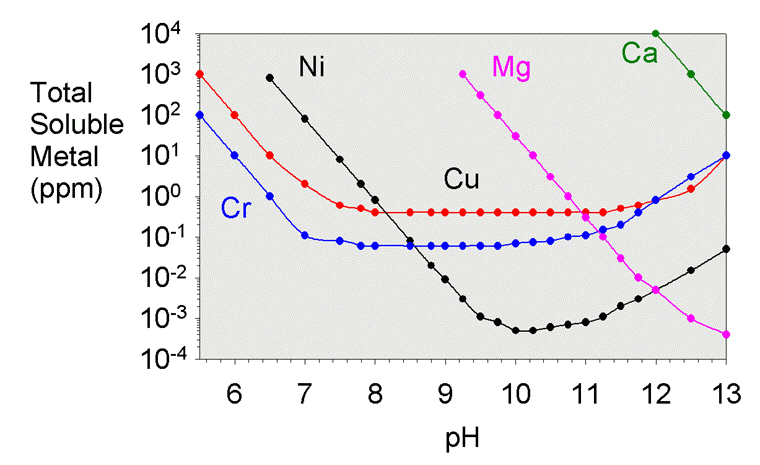
![Molar solubility of Ca (OH)2 in a solution that has a pH of 12. [ KSP [Ca (OH)2 ] = 5.6 × 10^- 12 ] Molar solubility of Ca (OH)2 in a solution that has a pH of 12. [ KSP [Ca (OH)2 ] = 5.6 × 10^- 12 ]](https://dwes9vv9u0550.cloudfront.net/images/10252669/718b1df9-54ff-4873-a48b-056b8accddcf.jpg)




![Mystery of solubility: Retrograde [Temperature Inverse] solubility: Why does it happen? Mystery of solubility: Retrograde [Temperature Inverse] solubility: Why does it happen?](https://media.licdn.com/dms/image/C5112AQHJlaA1CHtcdw/article-cover_image-shrink_600_2000/0/1520247821865?e=2147483647&v=beta&t=NDw0icvxf3iQGylvPgJCLn8uCwu0zFr38ky259eG9M0)